
Ficlatuzumab
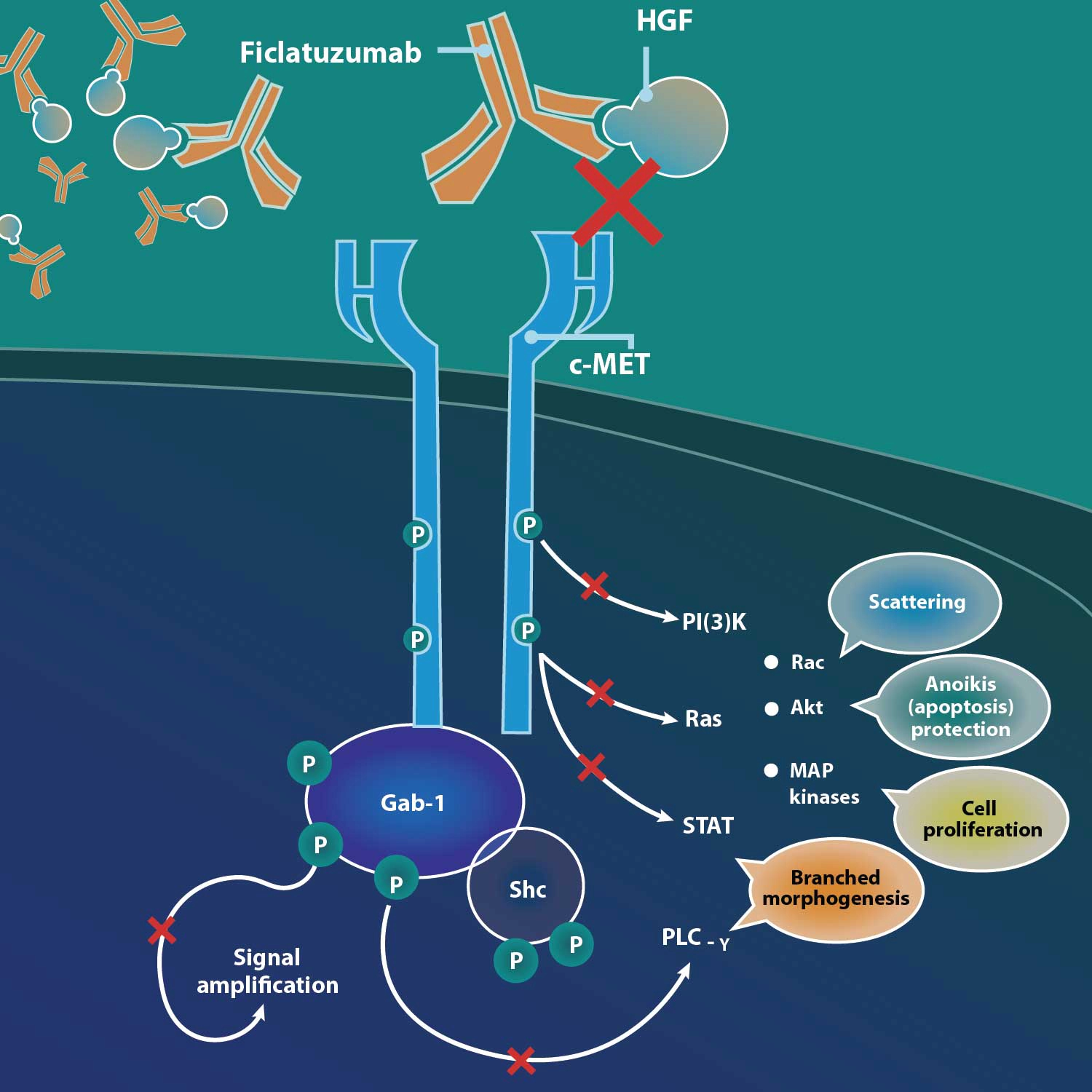
Ficlatuzumab (also known by research code AV-299) is a potent, humanized IgG1 monoclonal antibody that targets hepatocyte growth factor, or HGF. The HGF/cMET pathway is implicated as an escape mechanism for epidermal growth factor receptor, or EGFR, blockade. By binding to the HGF ligand with affinity and specificity, ficlatuzumab has demonstrated differentiated inhibition of HGF/cMET downstream signaling and demonstrated a strong additive anti-tumor effect in preclinical studies and early clinical trials.
HGF/cMET as a Therapeutic Target
HGF is the sole ligand for the cMET receptor. This pathway is frequently dysregulated in a broad range of human cancers. HGF binding with cMET can activate multiple intracellular signaling pathways and lead to both tumor growth and metastatic progression of cancer cells. HGF-induced MET activation is also an escape mechanism for EGFR inhibition, leading to resistance. Ficlatuzumab has demonstrated differentiated inhibition of HGF.
- High affinity (pM) and slow off-rate for HGF
- High potency (nM) inhibiting all biological activities of HGF, including autocrine/paracrine activation loops
AVEO has initiated a multicenter, randomized, double blind, placebo-controlled, phase 3 clinical trial of ficlatuzumab in combination with cetuximab in participants with recurrent or metastatic (R/M) HPV-negative head and neck squamous cell carcinoma (FIERCE-HN).
AVEO is currently enrolling patients in this study. For more information go to FIERCEHN.com or clinicaltrials.gov.
FIERCE-HN study design
Randomization
HPV-negative
R/M HNSCC
N≈410
Arm 1
IV ficlatuzumab dose A on Day 1 (D1) and D15 of each 28-day cycle IV cetuximab on D1 and D15 of each 28-day cycle
Arm 2
IV ficlatuzumab dose B on D1 and D15 of each 28-day cycle IV cetuximab on D1 and D15 of each 28-day cycle
Arm 3
IV placebo (saline) on D1 and D15 of each 28-day cycle IV cetuximab on D1 and D15 of each 28-day cycle
Ficlatuzumab in Other Indications
Ficlatuzumab has also been studied in combination with chemotherapy in pancreatic cancer and acute myeloid leukemia in early-stage clinical trials. Both studies demonstrated promising efficacy activity and an acceptable tolerability profile. AVEO is interested in novel combinations and continues to evaluate additional opportunities for exploring ficlatuzumab potential by actively seeking partners for clinical collaboration.
Ficlatuzumab is an investigational drug and has not yet been approved by the FDA.
Explore published ficlatuzumab clinical study data
AVEO has reported results from a randomized phase 2 clinical trial of ficlatuzumab with or without cetuximab (ERBITUX®) in patients with (pan-refractory) recurrent or metastatic HNSCC, including data related to progression-free survival and anti-tumor activity in patients who were HPV-negative randomized to the combination arm.
In development
As part of our goal to help patients with cancer live better lives, we are seeking to advance other candidates with the potential to positively impact treatment.
AVEO aims to leverage its existing collaborations and partnerships, as well as enter into new strategic collaborations and partnerships to continue to advance each of its product candidates.